| THERMO Spoken Here! ~ J. Pohl © | TOC ~ 245 |
Ideal Gas Elevator
The means of heating/cooling are not shown.
It is proposed to construct an elevator to operate in consequence of successive heating and cooling of air, the working substance. The apparatus will consist of a piston and cylinder. Figure 1 shows the intended elevator dimensions and specifications.
By inspection of the schematic we understand elevator operation will be five steps. A p-V diagram for the air is needed. States of the air are determined as the p-V diagram is sketched: Figure 2.

♦ Preheat: States (1) to (2): The elevator is charged with air initially in equilbrium with ambient surroundings (298K, 100kPa, 30m³). The piston mass and area are 230kg and 0.44m³. Therefore (by Newton's 2nd Law) the air must have the pressure p4 = 5,228 kPa before the piston will start to rise.
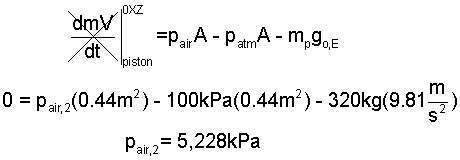
To calculate the heat (1) to (2) we require temperature, T2:
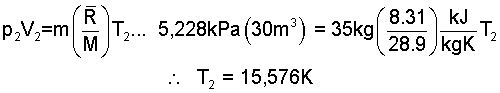
Preheat happens at constant volume hence work is zero. The energy equation is:
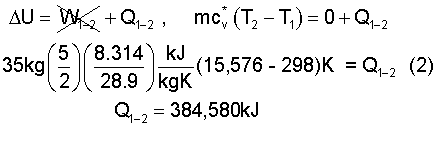
This brings us to State (2) (35kg, 30m³, 5,228kPa, 15,576K), the first state of intended cyclic operation of the elevator.
♦ Heat to Commence Lift: States (2) to (3) The air must attain pressure sufficient to lift the piston and casting. For pressure use the 2nd Law.
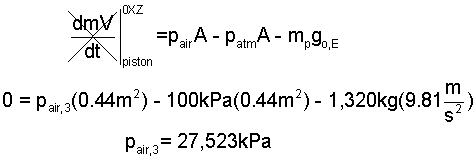
At state (3), assume air is an ideal gas so:
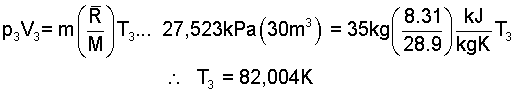
Heat the "Heat to Commence" is:

♦ Heat of Lift: States (3) to (4) The volume of air will increase to 32.64m³ as the air pressure remains constant. Thus we know the conditions of State (4) (30kg, 32.64³, 27,523kPa and T = T4. We might calculate W3-4 independently. However let's apply the energy equation and obtain work and heat together.

The return cycle has Steps, "Cool to commence movement downward ((4)-(5))" then "Cooling to move downward to Level 1 ((5)-(6)). These events, "reverses" of the above are not analyzed here.